Pluristem Therapeutic teams up with Japan’s Medical University to test radiation treatment on Fukushima victims
12/09/2015 / By Greg White

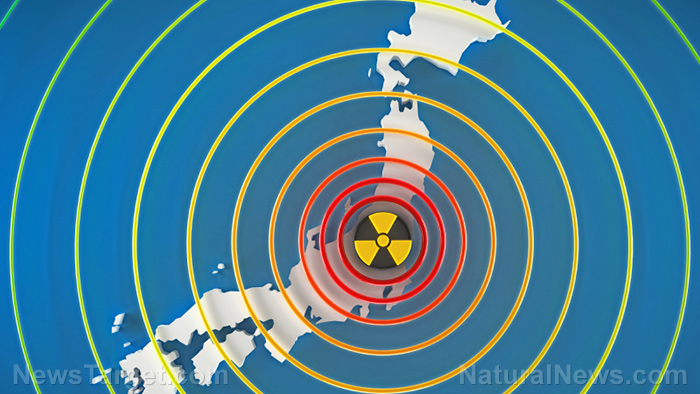
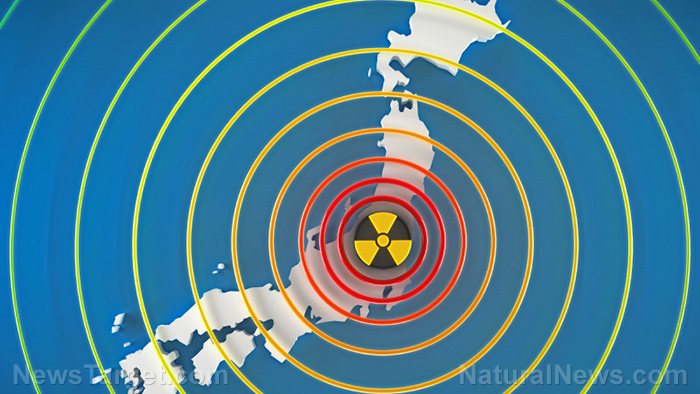
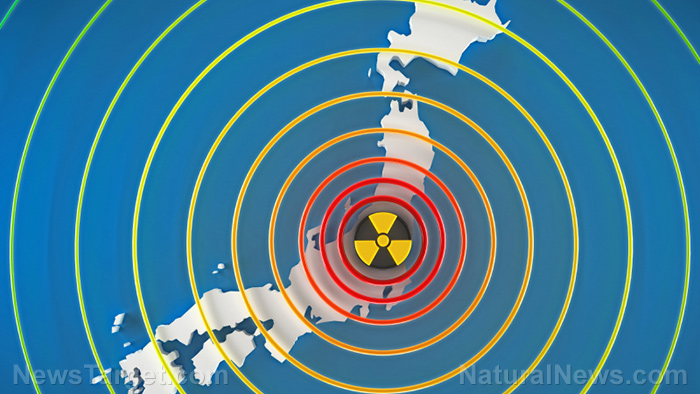
The Tepco Power Electric Company (TEPCO) has been trying to cover up the severity of the Fukushima Daiichi disaster for years. These efforts were called out for bluff in late October, after the Japanese government admitted nearly 40 percent of Defense Force troops, firefighters and police officers were exposed to radiation above the annual public limit of 1 millisievert. Consequently, Israeli biotech firm Pluristem Therapeutics have teamed with Japan’s Fukushima Medical University to test its radiation treatment derived from human placenta cells.
The purpose of the partnership is to create Pluristem’s PLX-R18 cells for the treatment of Acute Radiation Syndrome (ARS), and for abnormalities following radiotherapy in cancer patients. ARS is caused by exposure to toxic levels of radiation, which can damage the gastrointestinal tract, lung, skin, bone marrow and other vital organs.
Hundreds of deaths have been attributed to the Fukushima meltdown, both during and following the disaster. Nevertheless, in October, TEPCO finally acknowledged that there was a causal link between cancer and radiation exposure at the power plant among one of its employees. The organization has been struggling to keep the Fukushima site under control. It’s estimated that it will take decades and billion of dollars to decomission.(1)
Testing PLX-R18 to treat Acute Radiation Syndrome
The organizations involved in the study plan to develop pre-clinical trials of radiation damage on these tissues and test them during preliminary trials. Pluristem will provide the PLX-R18 cells and scientific background, while the Fukushima Medical University will conduct the trials and provide necessary resources. This joint collaboration will be conducted alongside the U.S. National Institutes of Health (NIH), which is studying how PLX-R18 could be used to treat ARS.
According to a preliminary study published in the journal PLOS One, PLX-R18 cells injected into the muscle may be a highly effective store-bought therapy for bone marrow failure following exposure to high levels of radiation.
“The researchers at Fukushima have a strong interest in cell therapies that may offer a treatment for conditions caused by exposure to ionizing radiation. We are honored to work with the thought leaders in the field at Fukushima Medical University, Fukushima Global Medical Science Center to further develop PLX-R18 for ARS-related indications,” reports Pluristem CEO, Zami Aberman.(2)
“We anticipate that our work with Fukushima Medical University, Fukushima Global Medical Science Center will help to maintain the health of those involved in decommissioning the Fukushima reactors; the decommissioning process is estimated to take about 40 years. We hope that this collaboration can advance efforts to safeguard populations around the world from illness related to radiation exposure,” he added.(2)
Fukushima: A goldmine for radiation therapy
Although TEPCO has publicly admitted that radiation bleeding from the power plant is linked to cancer, the situation is far worse than the company has led the masses to believe. A recent scientific study found a 30-fold excess of thyroid cancer among 400,000 young people below the age of 18 in the Fukushima Prefecture.
In the study, approximately 298,577 young people were screened four years after the disaster. Thyroid cancer rates were 50 times higher for young people in that group than for young people living outside the Fukushima Prefecture. In the second screening, 106,068 young people living in less contaminated regions in the Fukushima Prefecture were assessed. Among that group, cancer was 12 times more common than in the general population.(3)
Given these recent findings, it should be unsurprising that Pluristem Therapeutics has teamed up with Japan’s Fukushima Medical University to treat ARS. The Fukushima Prefecture is a goldmine for testing PLX-R18 cells on Japanese citizens exposed to dangerously high levels of radiation. Just don’t expect TECPO to acknowledge this fact. Otherwise, they would have more medical bills than they could possibly foot.
Sources include:
(1) Reuters.com
(2) Nasdaq.com
Submit a correction >>
Tagged Under:
AcuteRadiationSyndrome, FukushimaMedicalUniversity, PluristemTherapeutics
This article may contain statements that reflect the opinion of the author
RECENT NEWS & ARTICLES
COPYRIGHT © 2017 FUKUSHIMAWATCH.COM
All content posted on this site is protected under Free Speech. FukushimaWatch.com is not responsible for content written by contributing authors. The information on this site is provided for educational and entertainment purposes only. It is not intended as a substitute for professional advice of any kind. FukushimaWatch.com assumes no responsibility for the use or misuse of this material. All trademarks, registered trademarks and service marks mentioned on this site are the property of their respective owners.